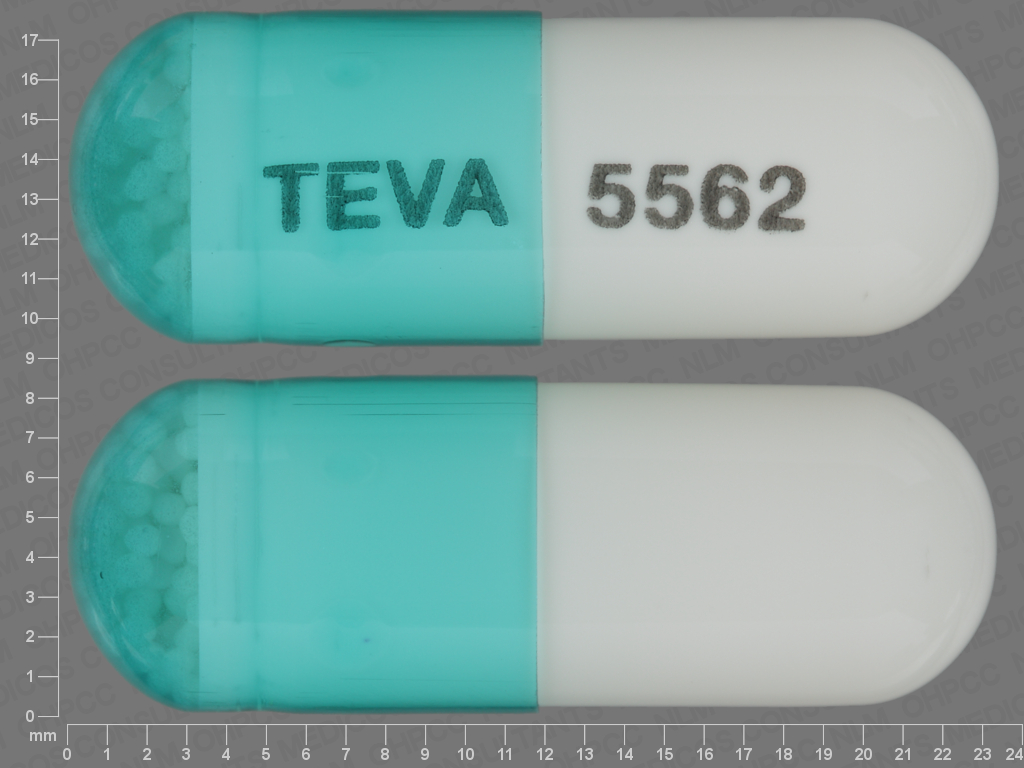
capsule teva 5562 Images - Dexmethylphenidate Hydrochloride - dexmethylphenidate hydrochloride - NDC ndc

Teva Parenteral Medicines Initiates Voluntary Nationwide Recall of Select Lots of Adrucil® (fluorouracil injection, USP) 5 g/100 mL (50 mg/mL) Due to Particulate Matter | Business Wire
Eli Lilly & Co. v. Teva Parenteral Medicines, Inc. (Fed. Cir. 2012) | McDonnell Boehnen Hulbert & Berghoff LLP - JDSupra











/cloudfront-us-east-2.images.arcpublishing.com/reuters/CCBS56SBRRKUDM7JEVVE3UOQEE.jpg)