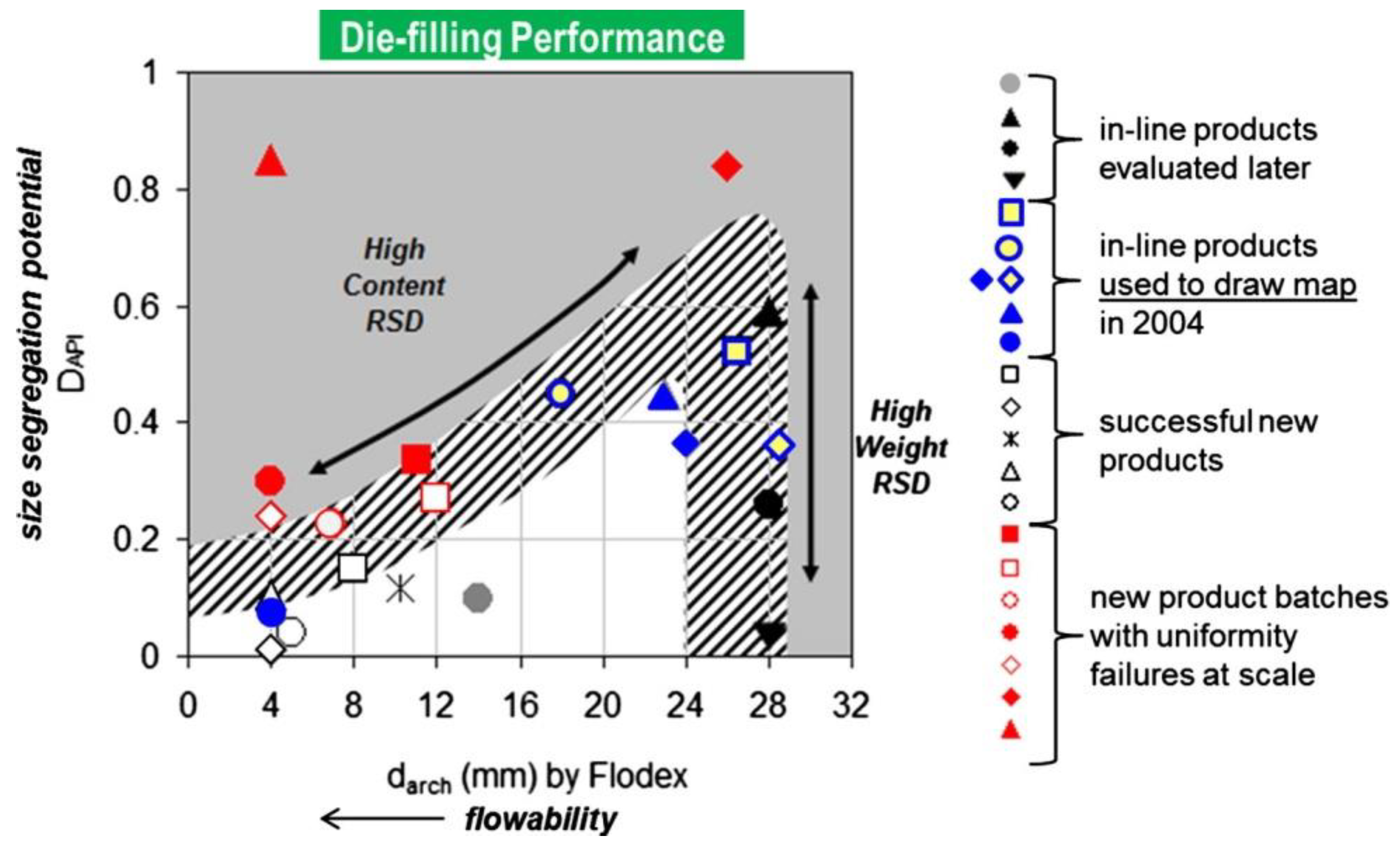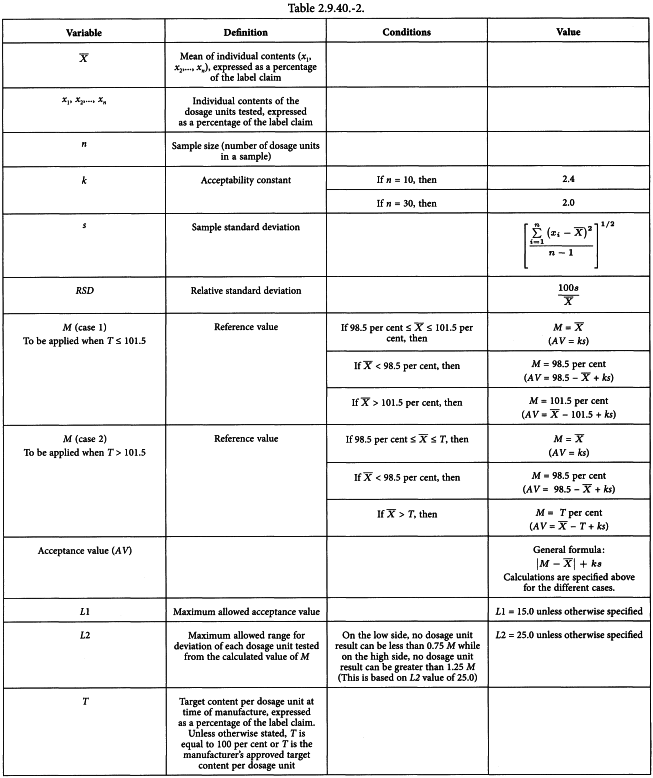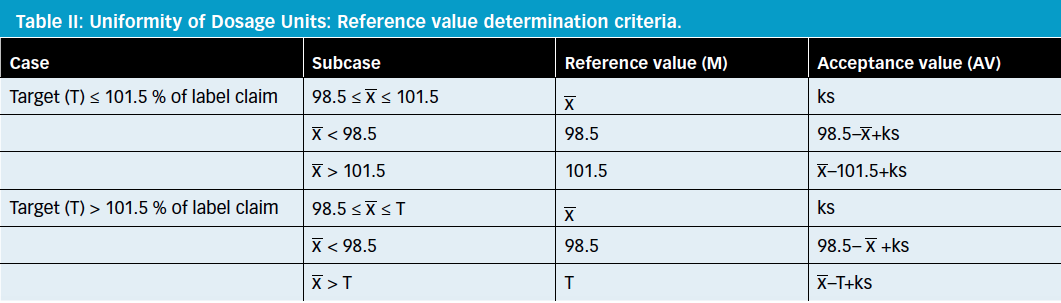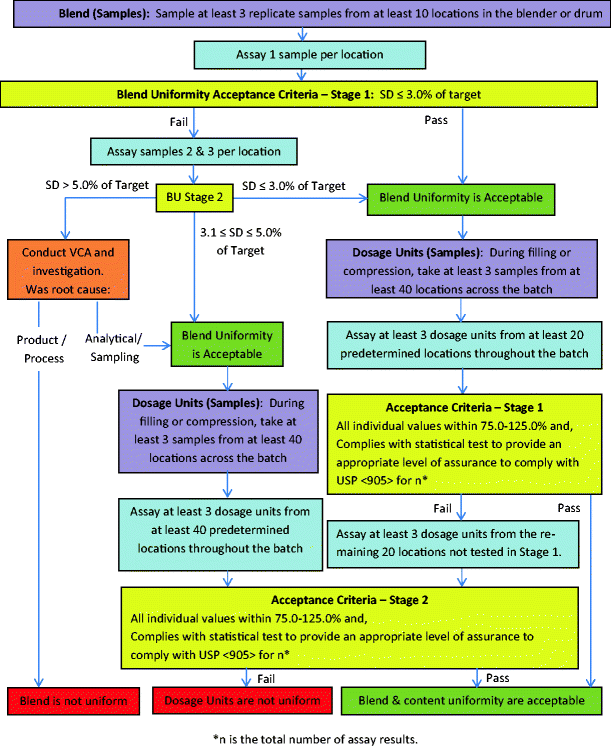
Recommendations for the Assessment of Blend and Content Uniformity: Modifications to Withdrawn FDA Draft Stratified Sampling Guidance | SpringerLink
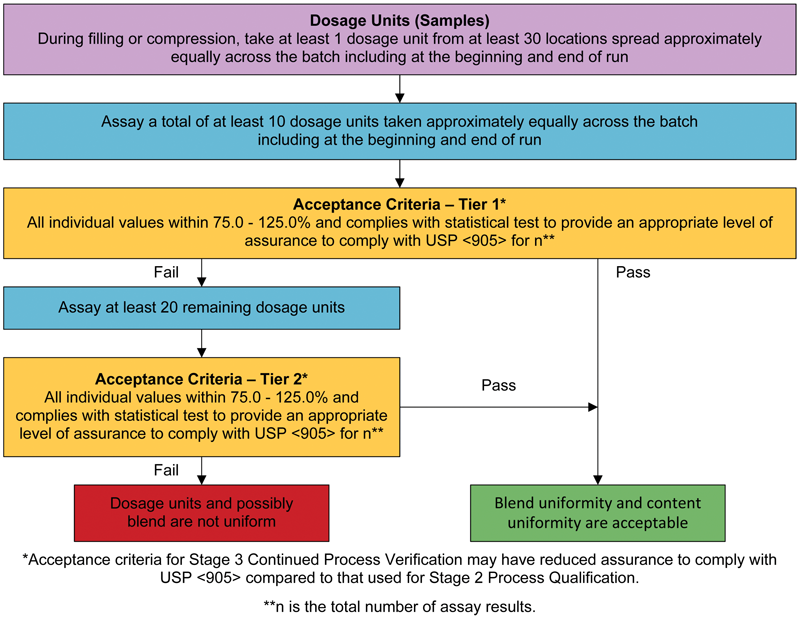
Comparison of Two Relevant Statistical Approaches to Assess Content Uniformity | Pharmaceutical Engineering

CONTENT UNIFORMITY TEST FOR TABLET DOSAGE FORM | EVALUATION PARAMETERS OF TABLET | PART -9 - YouTube

Content Uniformity Testing Through Utilization of Automated Dissolution Technologies | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

Scientific rationale for sampling regimen and acceptance criteria of blend uniformity based on Monte Carlo simulation - ScienceDirect

Analysis of weight uniformity, content uniformity and 30-day stability in halves and quarters of routinely prescribed cardiovascular medications. | Semantic Scholar
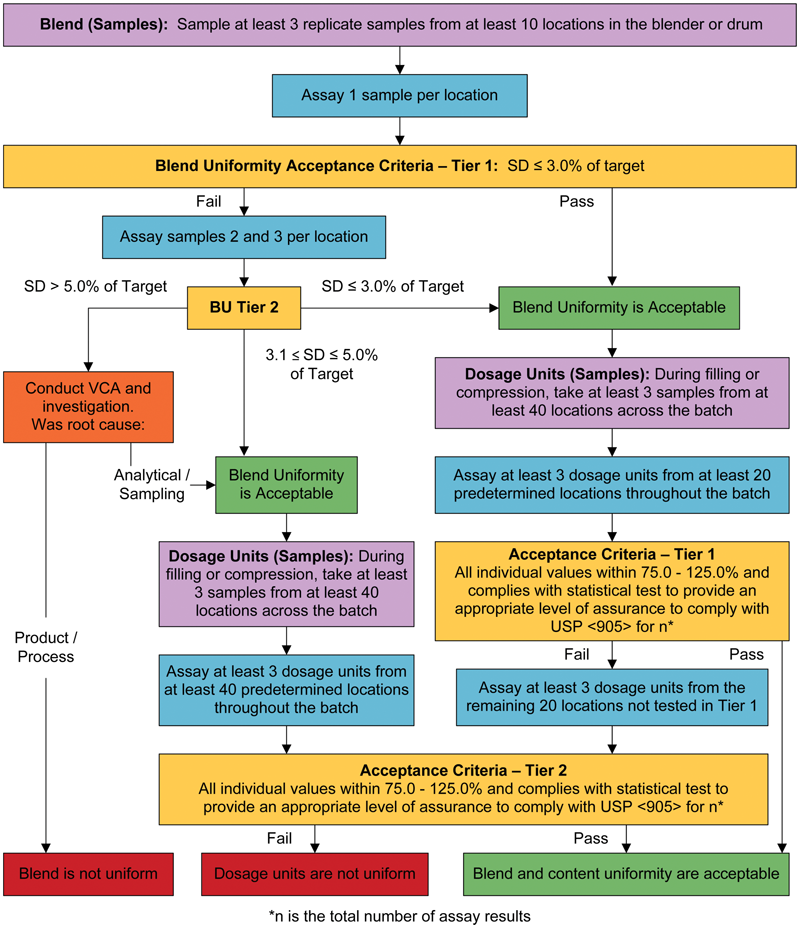
Comparison of Two Relevant Statistical Approaches to Assess Content Uniformity | Pharmaceutical Engineering